- Messages
- 32
- Reaction score
- 32
- Points
- 8
oh ok thanks , so the ideal gas equation can actually be used for any gas ?
yes it can be..
We are currently struggling to cover the operational costs of Xtremepapers, as a result we might have to shut this website down. Please donate if we have helped you and help make a difference in other students' lives!
Click here to Donate Now (View Announcement)
oh ok thanks , so the ideal gas equation can actually be used for any gas ?
9701_s09_qp_1.pdf
q 40 someone pleaseee explaainn!!
the bond will be broken from CO2 thats where the Na from NaOH will bond to oxygen.. it will not be 3 bcoz they have taken out the alkene completely so its wrong.. 2 is correct bcoz Na is bonded to O and the rest of the molecule is ok.. so definetly its going to be 1 also.. bcoz there is no option just for 2.. but u can still check it.. see if all the bonds are presnt or not and with OH which comes from NaOH
Thanks .. but can you explain it a bit more coz I still didn't understand why did the bond from CO2 break!
hmm :/
hmm :/
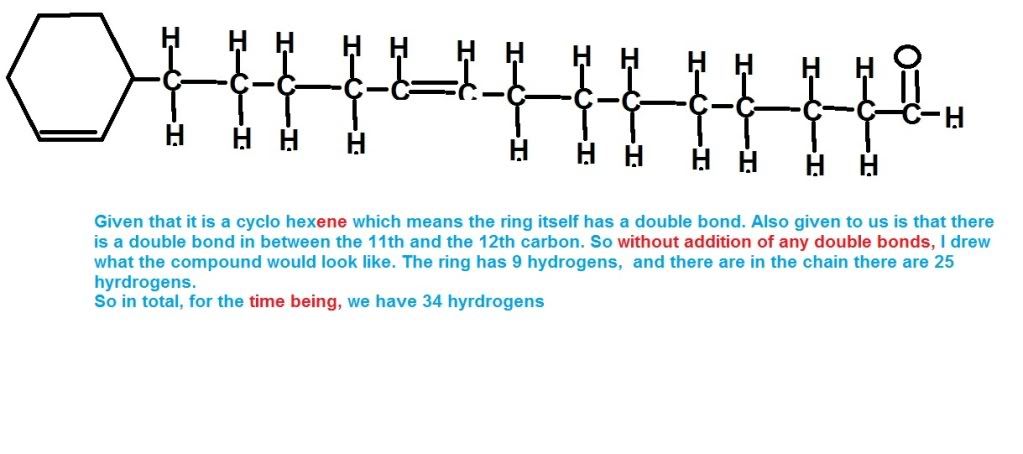
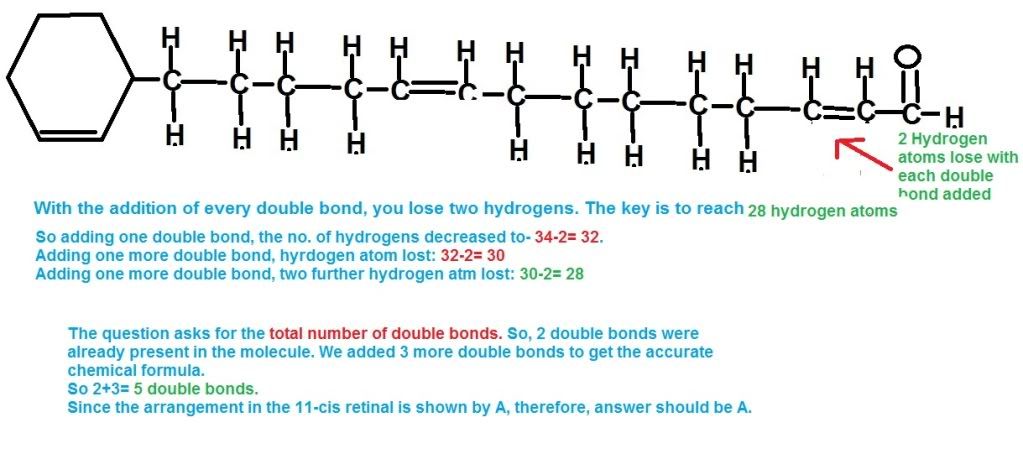
That question is f`d up.. totally went over my mind. Need help in this...
answer is B
amount of sulphite use = conc x vol
= 0.1 x (25/1000)
=2.5x10^-3mol
2 electrons are lost
amount of electrons lost = 2 x 2.5 x 10^-3 ==== 5 x 10^-3 mol
amount of electorns gained by metallic salt = amount of electrons lost by sulphite
= 5 x 10^-3 mol
amount of metallic salt used = 0.1 x (50/1000) ==== 5 x10^-3
amount of electrons gained PER MOLE of salt = (5 x 10^-3)/(5x10^-3) = 1 unit
hence oxidation state of metallic salt decreases by 1 unit meaning the oxidation satate becomes +2 from +3!!! this is for the question 9 , some other person posted it
i got it looksee da above post
no no on the left side it has O and on the right it has H in B part. in C it has O on one side and Cl on the other side. that is what i meant by OPPOSITE poles. thus dipoles are formed.Hay there,
Thanks for helping out, but its still not solved
Option B has H and H on both sides.. (not H and Cl, if that would been, then it would be a sure answer, but its not like that)
Option C has Cl and Cl on both sides..
And I'll be grateful if you explain the question number 10 of the same paper. Why its answer is C, whats wrong in B ??\
Regards
For more than 16 years, the site XtremePapers has been trying very hard to serve its users.
However, we are now struggling to cover its operational costs due to unforeseen circumstances. If we helped you in any way, kindly contribute and be the part of this effort. No act of kindness, no matter how small, is ever wasted.
Click here to Donate Now