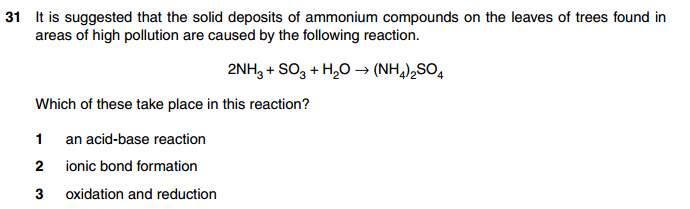- Messages
- 59
- Reaction score
- 130
- Points
- 43
http://papers.xtremepapers.com/CIE/... AS Level/Chemistry (9701)/9701_w10_qp_12.pdf
Number 7 and 8???? very hard . Anyone please explain them?
Number 7 and 8???? very hard . Anyone please explain them?