- Messages
- 1,247
- Reaction score
- 2,326
- Points
- 273
I dont understand this logic?
How did this clicked to ur mind?
I don't know how else to explain it. Sorry.
We are currently struggling to cover the operational costs of Xtremepapers, as a result we might have to shut this website down. Please donate if we have helped you and help make a difference in other students' lives!
Click here to Donate Now (View Announcement)
I dont understand this logic?
How did this clicked to ur mind?
He might have said it in a bad way, the "period" he found by dividing 0.45/0.3 is the number of cycles in 45cm of lineI dont understand this logic?
How did this clicked to ur mind?
I figured it out something like this, There are two nodes formed at transmitter and at plate. So that is lambda/2 = 0.45. Hence lambda = 0.9m. Also I did, lambda = 3*10^8 / 1 * 10 ^9 = 0.3m.He might have said it in a bad way, the "period" he found by dividing 0.45/0.3 is the number of cycles in 45cm of line
so 1 cycle= 0.3m
how many cycles in 0.45 m?
you just divide and get 1.5 cycles.
Which equals to 1 complete cycle and 1/2 a cycle
Just draw , you understand it right away
No, lambda/2 is used for adjacent nodes. The two nodes in the question are not adjacent.I figured it out something like this, There are two nodes formed at transmitter and at plate. So that is lambda/2 = 0.45. Hence lambda = 0.9m. Also I did, lambda = 3*10^8 / 1 * 10 ^9 = 0.3m.
Therefore 1 antinode in 0.3m so how many in 0.9?
Hence answer is 3.
Is it correct?
Thanks.
So its wrong...No, lambda/2 is used for adjacent nodes. The two nodes in the question are not adjacent.
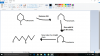
Okay, first, you have to realize that X cannot be a cyclic compound. Why? Because the NH group in coniine is the reason the compound is cyclic. In its absence, the compound is a straight chain. Now notice one thing. When 'N' is removed, that leaves two ends, which is originally occupied by the two 'Br' atoms. When you draw X as a straight chain, you'll notice that the 1st Br atom attaches to the first carbon, and the 2nd Br attaches to the fifth carbon. Therefore, the compound is 1,5-dibromooctane.
26 for Physics. Dunno about Chem.my avg score for chem is 31 and for phy is 29
what about u all???
From which paper is this question from?
I dont rememberFrom which paper is this question from?
Oh..okeyI dont remember
There is same functional groups, so there would be one same cis trans isomer hence 3View attachment 57338
For q29 i chose D don't remember why but the answer is C
I drew the structures..i just wanna know if they are correct
View attachment 57338 View attachment 57339
I know this questionView attachment 57340
Ans is C..how?
Lol okeyI know this question
I will have my dinner and solve it
I am in hostel
I totally forgot.. :3Lol okey
For more than 16 years, the site XtremePapers has been trying very hard to serve its users.
However, we are now struggling to cover its operational costs due to unforeseen circumstances. If we helped you in any way, kindly contribute and be the part of this effort. No act of kindness, no matter how small, is ever wasted.
Click here to Donate Now